Work by Kyle Parcher
A Portland State University physics student
A Portland State University physics student
Applied physics in biology: mapping the interior of membranes
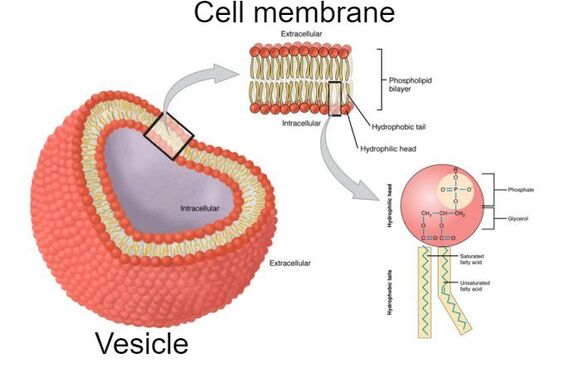
Composition of the cell membrane
To begin, lets look at the vesicle. The vesicle is a closed shape, comprised of the cell membrane and the contents inside of it. The cell membrane is what keeps everything together. It is vital to cell function and can be presented in many different variations. The cell membrane itself is made of a phospholipid bilayer. This bilayer is formed due to the repulsion of water. If we look at one side of the bilayer, or rather the phospholipid itself, we see that one side repels water, the either attracts it. Since cells are invariably in an aqueous solution, the bilayer forms.
|
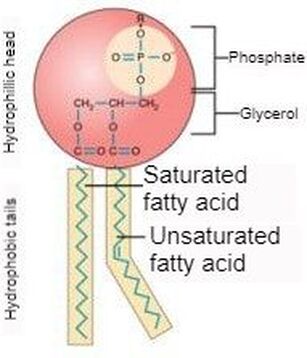
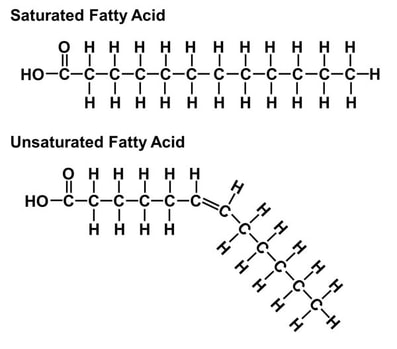
Taking a closer look at the phospholipid, we see that the head (colored in red) is the phosphate group while the tail is the grease group (depicted within the yellow of the diagram). One must understand that grease is a hydrocarbon chain that can be considered a saturated fatty acid chain, or a unsaturated one. One can also think of these groups just as saturated and unsaturated fats. The difference? The absence or presence of double bonds between carbon groups. |
Saturated vs. unsaturated fats
|
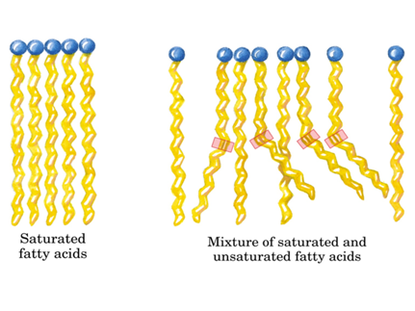
As stated above, the difference is the absence or presence of double bonds between each carbon. A saturated fatty acid has no double bonds while an unsaturated fatty acid has at least one (shown to the right). When an unsaturated fatty acid has more than one double bond between carbon groups, it is referred to as a poly unsaturated fat. An advantage an unsaturated cell membrane has over a saturated one is a lower bilayer density. When there is a lower density bilayer, there is an easier passage of nutrients into the cell. This difference of bilayer structure is shown below and to the right. The importance of this difference (to cell structure and to ones health) becomes apparent when we examine the phase difference between a stick of butter and olive oil at room temperature. If a cell membrane is comprised of grease similar to butter and is at a temperature below the freezing point of butter, the passage of nutrients is extremely restricted due to the bilayer becoming solid. For a real world example, we can examine fish that reside in arctic regions. For these fish, having a bilayer with a melting point of olive oil would not suffice either. It just so happens that fish oil is a polyunsaturated fatty acid, made almost elusively with double bonds between carbon groups. Most importantly, it has a very low melting temperature. This is vital to the survival of this creature for its cells don't solidify and block the passage of nutrients into cells.
|
FLUORESCENCE and cell membranes
|
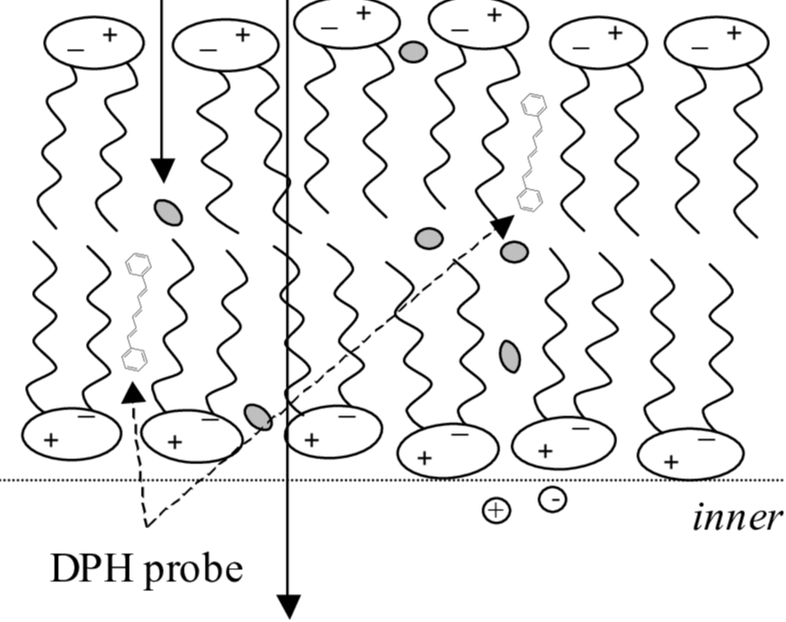
Through a nontrivial process, a sample can be prepared of cell membranes that can come in various different flavors. For the most part, these flavors (amount of double bonds) can be broken into two groups: saturated and unsaturated. These samples are in an aqueous solution where they are brought to a fluoroscope. The fluoroscope begins to measure when we introduce out fluorescent probe. This reaction is on a scale that is difficult to fathom. The cell membranes that are being mapped are smaller than a micrometer (a millionth of a meter) using fluorescence that lasts nanoseconds (billionth of a second). Some methods can last much longer to measure movement, such as the GIF to the right.
|
The mapping of cell membranes is done via fluorescent probes inserted into the cell membrane. This process is called extrusion. When the probe is inserted, certain movements can be measured. This is two translational and one rotational movements. Based on these movements, the fluoroscope picks up more or less of the fluorescence emitted based on the probes orientation. Since we can pick the type of cell membrane we work with, we can measure how the probe interacts with the membrane in terms of how it moves within it. Due to the way the probe moves, we can reason some things about how it would move within other cell membranes. Additionally, movement is not the only we can measure. We can measure lifetime of the probe. Based on what we can measure, we can learn about cell membrane structure and do some mapping with fluorescence. |
This work is important because we can look into the effects of diet on a molecular level. We are not limited to the effects of diet either. This practice can be applied to how pharmaceuticals interact with the body, or how different cell structures are affected by temperature. As of writing this, I am planning on researching the effects of Taxol, a chemotherapy drug designed to prevent cancer cells from dividing, on the human body. A big issue with these chemotherapy drugs is that, while it destroys the cancer cells, it also destroys some of the body with it. Treatment with Taxol is dependent on the number of platelets in the body. A patients next treatment is contingent on their body healing enough (producing enough platelets) from the prior dose of Taxol.
About the student
|
Kyle Parcher is a general physics major and math minor at Portland State University at the time of publication of this website. He is 23 years old at the date of writing and will graduate Portland State University at the end of the 2019 - 2020 school year. He plans to start a career in physics, but he has not yet narrowed down his field of study.
|
About the professor
|
Dr. Drake Mitchell is a professor of physics at Portland State University. He obtained his Ph. D. in physics at the University of Oregon in 1987. He now conducts research in the field of biophysics where his research interests and technical specialties lie in fluorescence, calorimetry, liposomes and biological membrane mimetics.
|